Applied Quantum Systems
News
Department
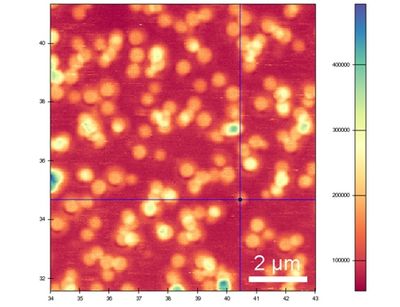
Main goal of the AQS group is to fabricate quantum devices on solid state basis and develop new methods to fulfil this task. Our technology of choice is ion beam implantation. Ions with kinetic energies allow the three dimensional modification of all types of solid state materials.
Research
Research projects, publications and innovations spring again and again from the impressive work in our department, supported by first-class laboratory equipment.
Study
Here, students get a first insight into the spheres of physics, its learning content and teaching objectives, as well as the special features of studying at our department.
Contact